Category- Allopathy
Medicine Highlights
| Uses | 1. To reduce pressure on eyes in glaucoma |
| Common Side Effects | 1. Body aches or pain 2. Cough 3. Difficulty with breathing 4. Ear congestion 5. Loss of voice 6. Redness of the white part of eyes or inside of the eyelids 7. Sneezing 8. Sore throat 9. Stuffy or runny nose 10. Unusual tiredness or weakness |
| Additional Information | 1. Avoid touching the tip of the container. 2. Avoid using the drops if the seal is broken. |
| Storage | 1. The medicine should be stored at room temperature. 2. Keep away from sunlight and moisture. 3. Keep away from the reach of children |
| How to Take This Medicine | Instill one or two drops into the eye pocket without touching the bottle tip. |
Travoprost Uses
Travoprost 0.004% is an oral medicine belonging to the category of prostaglandin analogues and is used for the management of glaucoma in eyes.
Travoprost side effects
More Common
- Body aches or pain
- Cough
- Difficulty with breathing
- Ear congestion
- Loss of voice
- Redness of the white part of eyes or inside of the eyelids
- Sneezing
- Sore throat
- Stuffy or runny nose
- Unusual tiredness or weakness
Less Common
- Blindness
- Bloody eye
- Blurred or decreased vision
- Change in color vision
- Color changes in the skin around the eyes
- Difficulty seeing at night
- Disturbed color perception
- Double vision
- Dry eyes
- Eye color changes
- Fever or chills
- Halos around lights
- Lack or loss of strength
- Loss of vision
- Night blindness
- Overbright appearance of lights
- Redness, burning, dry, or itching eyes
- Redness, pain, swelling of the eye, eyelid, or inner lining of the eyelid
- Tunnel vision
How Travoprost works
Travoprost works by helping fluid inside the eye drain more easily, which lowers eye pressure. Lower pressure protects the optic nerve and helps prevent vision loss from glaucoma. It can also make eyelashes grow longer, thicker, and darker when applied to the eyelid.
Safety Measures and Warning
Pregnancy
Travoprost use in pregnancy is generally avoided due to the limited evidence related to safety in pregnancy. Always consult a doctor before using it.
Breastfeeding
Travoprost enters breast milk in small amounts, and it is safe for use in breastfeeding, but caution is advised, and it should be used only if clearly needed under a doctor’s recommendation.
Eye Infections
Patients with active eye infections should avoid travoprost because the drops can worsen irritation and inflammation in already compromised tissues. Always consult a doctor before using it.
Travoprost Interaction with Food Product
Avoid consuming alcohol and smoking simultaneously, as it may affect the drug’s action and show an effect on the brain, leading to severe drowsiness, sedation, and coma in severe cases.
Travoprost Interaction with Other Medicines
Avoid taking Travoprost 0.004% with the following medicines, as it may increase or decrease the desired effect, or in worse cases, it could be fatal:
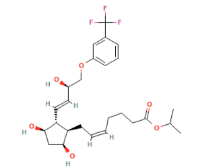
Travoprost Chemical Structure
IUPAC (International Union of Pure and Applied Chemistry)/ Chemical name
propan-2-yl (Z)-7-[(1R,2R,3R,5S)-3,5-dihydroxy-2-[(E,3R)-3-hydroxy-4-[3-(trifluoromethyl)phenoxy]but-1-enyl]cyclopentyl]hept-5-enoate
Intake routine/cycle
Daily dose
Take Travoprost exactly as your doctor recommends. The dose may change based on your condition and how well you respond. Do not take more than prescribed without consulting your doctor.
Missed Dose
If you forget to take your medicine, take the medication as soon as you remember. If it’s nearly time for your next dose, skip the missed one and continue with your regular schedule. Always consult the doctor before making any changes to your dosage.
Overdose
It is not recommended to use more than 6 drops of Travoprost per day, as it may cause severe overdosing effects like dry mouth, stomach upset, and irritation. Immediately seek medical attention and meet the doctor if necessary.
Storage
- Store the medicine at room temperature.
- Keep away from sunlight and moisture.
- Keep out of reach of children.
Directions for use
- Take Travoprost exactly as prescribed by your doctor, at the same time each day.
- Do not stop taking Travoprost without your doctor’s approval, even if you feel better.
- If you forget to take Travoprost, take it as soon as you remember. If it’s almost time for your next dose, skip the missed dose.
Travoprost Summary
Travoprost is a prostaglandin analog used for reducing high eye pressure in glaucoma and ocular hypertension and cosmetically for increasing eyelash growth. It works by enhancing the outflow of fluid from the eye, thereby lowering pressure in the eyes. Common side effects include eye redness, itching, dryness, and darkening of the eyelid skin or iris. Taking travoprost with other eye drops without medical advice can increase irritation or reduce effectiveness. People with eye infections, uveitis, or prior eye surgery complications should avoid it. Overuse may cause permanent color change of the iris, excessive eyelash growth, and vision disturbances.
FAQ’s
What are the uses of Travoprost eye drops?
Travoprost ophthalmic drops are primarily used to lower intraocular pressure in patients with open-angle glaucoma or ocular hypertension, and to treat eyelash hypotrichosis by promoting eyelash growth
What are the side effects of Travoprost drops?
Travoprost eye drops can cause common side effects like eye redness, itching, eyelid darkening, and eyelash growth, while rare but serious risks include iris color changes and macular edema. Most effects are localized to the eye, with systemic side effects being very uncommon.
What is the drug class of Travoprost?
Travoprost belongs to the drug class of prostaglandin analogues.
What are Travoprost interactions with other drugs?
Travoprost eye drops mainly interact with other prostaglandin analogs (like latanoprost, travoprost, tafluprost), where combined use can reduce effectiveness or increase side effects. It has few systemic drug interactions due to minimal absorption, but caution is advised in patients with certain eye conditions.
Reference
- StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan–. Prostaglandins Medications. Available from.
- National Center for Biotechnology Information (NCBI). PubChem Compound Summary for CID 5282438, Travoprost [Internet]. Bethesda (MD): National Library of Medicine (US); [cited 2026 Jan 29]. Available from.
- Drugs.com. Travoprost (Professional Patient Advice) [Internet]. Drugs.com; [cited 2026 Jan 29]. Available from.
- Mayo Clinic. Travoprost (ophthalmic route) [Internet]. Rochester (MN): Mayo Foundation for Medical Education and Research; [cited 2026 Jan 29]. Available from.

Siva Krishna Adithya Bhumireddy completed a Bachelor of Pharmacy from GIET School of Pharmacy, affiliated with Andhra University, and from his 3rd year of graduation, he was working as a freelance medical and publication writer. Having expertise in systematic reviews and clinical research. With his keen interest and skills in research and documentation practices, he also reviewed more than 150 manuscripts from international journals like Elsevier, Springer Nature, and Sage Journals, and all the manuscripts were indexed and appeared in PubMed, Scopus, and Web of Science databases, and he received honorary recognition letters and certificates from all of them. Siva Krishna Adithya also published a few systematic, narrative reviews and a few chapters of international expertise.
