Category- Allopathy
Medicine Highlights
| Uses | 1. To treat seizures (petit mal and unlocalized type) 2. To treat mountain sickness 3. To reduce pressure on eyes in glaucoma 4. To treat fluid retention in body |
| Common Side Effects | 1. Diarrhea 2. Vomiting 3. Loss of appetite 4. Numbness or tingling in arms or legs 5. Increased thirst 6. Increased urination frequency 7. Drowsiness 8. Headache 9. Change in taste/Metallic taste 10. Decrease in sexual drive |
| Additional Information | 1. Take it before the meals to get the maximum effect. 2. As this is a prescription medicine, use this medicine with a doctor’s recommendation. |
| Storage | 1. The medicine should be stored at room temperature. 2. Keep away from sunlight and moisture. 3. Keep away from the reach of children |
| How to Take This Medicine | Take this medicine orally with a sip of water. |
BrandName Uses
BrandName is an oral medicine belonging to the category of carbonic anhydrase inhibitors and is used for the following indications:
- Seizures
- Mountain Sickness
- Glaucoma
- Fluid retention in body
BrandName side effects
More Common
- Diarrhea
- Vomiting
- Loss of appetite
- Numbness or tingling in arms or legs
- Increased thirst
- Increased urination frequency
- Drowsiness
- Headache
- Change in taste/Metallic taste
- Decrease in sexual drive
Less Common
- Confusion
- Blood in urine
- Yellowing of skin and eyes
- Ringing or buzzing in the ears Severe skin rash and hives
How BrandName works
BrandName works by slowing down an enzyme called carbonic anhydrase inhibitor in the body that normally helps make and move fluids. By blocking this enzyme, it reduces the buildup of fluid and pressure in places like the eyes and brain. This helps lower eye pressure in glaucoma, prevent altitude sickness, and ease swelling or fluid retention.
Safety Measures and Warning
Pregnancy
BrandName use in pregnancy is generally avoided due to the possible risk of birth defects and poor fetal growth, though it may be considered only for serious conditions when safer options fail. Always take it strictly under a doctor’s recommendation.
Breastfeeding
BrandName enters breast milk in small amounts, and it is safe for use in breastfeeding, but caution is advised, and it should be used only if clearly needed under a doctor’s recommendation.
Kidney and Liver Problems
BrandName should be avoided in severe kidney or liver problems because it can worsen electrolyte imbalance and increase the risk of toxic buildup. In kidney disease, it may cause dangerous acidosis. Always consult a doctor before using it.
BrandName Interaction with Food Product
Avoid consuming alcohol and smoking simultaneously, as it may affect the drug’s action and show an effect on the brain, leading to severe drowsiness, sedation, and coma in severe cases.
BrandName Interaction with Other Medicines
Avoid taking BrandName with the following medicines, as it may increase or decrease the desired effect, or in worse cases, it could be fatal:
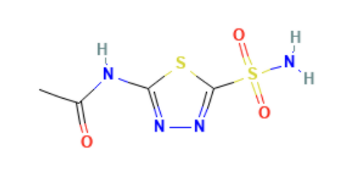
BrandName Chemical Structure
IUPAC (International Union of Pure and Applied Chemistry)/ Chemical name
N-(5-sulfamoyl-1,3,4-thiadiazol-2-yl)acetamide.
Intake routine/cycle
Daily dose
Take BrandName exactly as your doctor recommends. The dose may change based on your condition and how well you respond. Do not take more than prescribed without consulting your doctor.
Missed Dose
If you forget to take your medicine, take the medication as soon as you remember. If it’s nearly time for your next dose, skip the missed one and continue with your regular schedule. Always consult the doctor before making any changes to your dosage.
Overdose
It is not recommended to take more than 1000 mg of BrandName per day, as it may cause severe overdosing effects like extreme drowsiness, heart problems, confusion, unconsciousness, sweating, double vision, low blood pressure, and slurred speech. Immediately seek medical attention and meet the doctor if necessary.
Storage
- Store the medicine at room temperature.
- Keep away from sunlight and moisture.
- Keep out of reach of children.
Directions for use
- Take BrandName exactly as prescribed by your doctor, at the same time each day.
- Do not stop taking BrandName without your doctor’s approval, even if you feel better.
- If you forget to take BrandName, take it as soon as you remember. If it’s almost time for your next dose, skip the missed dose.
BrandName Summary
BrandName is a medicine that improves the gut motility and reduces stomach discomfort. It belongs to the prokinetic drug class and is not FDA approved but is used in some countries for reflux disease and indigestion. Its off-label uses are gastroparesis, constipation, and irritable bowel syndrome. Common side effects include diarrhea, abdominal pain, headache, drowsiness, and nausea. It works by stimulating serotonin receptors and blocking dopamine to speed up digestion. Food does not cause major interactions, though alcohol and sedatives can worsen drowsiness. Vulnerable groups include pregnant women, breastfeeding mothers, children, and patients with severe liver or kidney disease. Overdoses above about 100 mg may cause severe diarrhea, abdominal cramps, dizziness, tremors, and nervous system symptoms.
FAQ’s
What are the uses of BrandName tablets?
BrandName tablets are used to lower eye pressure in glaucoma, prevent altitude sickness, and treat fluid retention or certain types of epilepsy. They work by reducing excess fluid buildup and balancing pressure in the body.
What are the side effects of BrandName tablets?
BrandName tablets can cause common side effects like tingling in hands/feet, frequent urination, nausea, and fatigue; serious risks include electrolyte imbalance, kidney stones, skin reactions, and confusion, so medical monitoring is essential.
What is the drug class of BrandName?
BrandName belongs to the drug class of carbonic anhydrase inhibitors.
What are BrandName interactions with other drugs?
BrandName interacts with many drugs, especially diuretics, aspirin, lithium, and seizure medicines; these interactions can increase risks like electrolyte imbalance, toxicity, or reduced drug effectiveness, so it should only be combined under medical supervision.
Reference
- National Center for Biotechnology Information. PubChem Compound Summary for CID 1986, Acetazolamide. PubChem [Internet]. Available from.
- Mayo Clinic. Acetazolamide (oral route) – Description and Precautions. Mayo Clinic [Internet]. Available from.
- Drugs.com. Acetazolamide: Uses, Dosage, Side Effects. Drugs.com [Internet]. Available from.

Siva Krishna Adithya Bhumireddy completed a Bachelor of Pharmacy from GIET School of Pharmacy, affiliated with Andhra University, and from his 3rd year of graduation, he was working as a freelance medical and publication writer. Having expertise in systematic reviews and clinical research. With his keen interest and skills in research and documentation practices, he also reviewed more than 150 manuscripts from international journals like Elsevier, Springer Nature, and Sage Journals, and all the manuscripts were indexed and appeared in PubMed, Scopus, and Web of Science databases, and he received honorary recognition letters and certificates from all of them. Siva Krishna Adithya also published a few systematic, narrative reviews and a few chapters of international expertise.
